Prevenar 13
Most vaccines are non-live, and the vaccinated person does not affect others in any way. Some vaccines contain live, weakened pathogens that the vaccinated person can release into the environment. These weakened pathogens (viruses) are not dangerous for the vast majority, but caution should be used in some situations. The greatest number of restrictions exist for the live oral polio vaccine (OPV), since the vaccinated person releases the vaccine virus into the environment within 60 days:
1. Close contact of vaccinated children with children completely unvaccinated against polio is undesirable due to the risk of vaccine-associated polio in susceptible individuals. For the same reason, when vaccinating against polio, the first two doses are given with a non-live vaccine. Playing in the sandbox, walking together, and even visiting are not close contact! Transmission of the vaccine virus occurs through the fecal-oral route, so compliance with hygiene rules mitigates the risks.
2. Vaccination with OPV of older children, if the mother is pregnant and in late pregnancy, is not advisable, since separation of the older child and an unvaccinated newborn can be problematic. In this case, it is better to postpone OPV vaccination until the baby has received 2 doses of inactivated polio vaccine (IPV) and give OPV to both children at the same time, or, if possible, replace it with IPV.
3. Adults and children in a state of immunosuppression should also avoid close contact with people vaccinated with OPV.
The total risk of VAPP for unvaccinated contacts and for children who received OPV as the first dose is 2-4 cases per 1,000,000. As a rule, we are talking about children with immunodeficiency conditions, which themselves are very rare.
A person vaccinated with an oral vaccine against rotavirus infection can also release the vaccine virus into the environment, but it is not dangerous for contacts.
When living together with people in a state of immunosuppression, hygiene measures should be observed, since the vaccine virus can cause gastroenteritis in them.
A person vaccinated against chickenpox may shed the vaccine virus if they develop a rash as a reaction to the vaccine. Direct contact with the rash is necessary for transmission of the virus. It is recommended that it be avoided by unvaccinated and previously disease-free people who are in a state of immunosuppression, as well as non-immune pregnant women. For everyone else, contact with a vaccinated person is completely safe!
Vaccinated with live vaccines against measles, rubella, mumps and tuberculosis are not dangerous for others with any immune status!
More details
When to see a doctor
In medicine, there are three degrees of severity of redness and induration that appear after a child’s vaccination. Let's look at them:
- Mild degree - manifestations do not exceed 2.5 cm in size;
- Medium degree - size within 2.5-5 cm;
- Severe degree – more than 5 cm.
Manifestations after vaccinations are a normal response of the body to most inactivated drugs. Minor redness or swelling is not a reason for parents to raise the alarm. Give your child the necessary attention and wait out this moment.
Popular Flu will not go away when prevented with the Inflexal V vaccine
If parents observe residual symptoms in the child for more than a week after vaccination, and the size of the bumps or redness is 5 cm or more, it is necessary to consult a doctor without delay.
Possible side effects
In children 6 weeks to 17 years of age, the most common side effects are irritation, redness or swelling at the injection site, irritability, decreased appetite, decreased or increased sleep, and fever.
Serious but very rare side effects in infants and toddlers include pneumonia, bronchiolitis and gastroenteritis (inflammation of the stomach and small intestine) (0.9% of all vaccine recipients). A temporary pause in breathing after vaccination occurs in some babies born prematurely.
The high safety of vaccination has been confirmed by 230 million years of experience in administering Prevenar over the past 10 years. The vaccine is registered in 88 countries and is included in the national vaccination calendars of 30 countries.
Come get vaccinated at ONNI.
A full range of vaccines for children and adults, family vaccinations - at a special price! Call a doctor at home Make an appointment with a doctor or call +7 (812) 331-17-74
Vaccine Prevenar
7-valent pneumococcal polysaccharide conjugated adsorbed vaccine for the prevention of diseases caused by Streptococcus pneumoniae
,
Composition: each dose of the Prevenar vaccine contains purified polysaccharides of pneumococci of 7 serotypes, individually conjugated to the diphtheria carrier protein CRM197, and adsorbed on aluminum phosphate.
Release form: 0.5 ml of the drug in disposable syringes made of transparent, colorless borosilicate glass.
Indicated for use: for the prevention of diseases caused by Streptococcus pneumoniae serotypes 4, 6B, 9V, 14, 18C, 19F and 23F (including sepsis, meningitis, pneumonia, bacteremia and acute otitis media) in children aged 2 months to 5 years.
Vaccination schedule:
- children aged 2 to 6 months . according to the 3 + 1 revaccination scheme: 3 doses with an interval of at least 1 month, 1 dose is usually administered at the age of 2 months, the 4th dose (booster) is recommended to be administered at the 2nd year of life, optimally at 12-15 months;
- children aged 7 to 11 months . according to the 2 + 1 revaccination scheme: 2 doses with an interval of at least 1 month, the 3rd dose is recommended to be administered at the 2nd year of life;
- children aged 12 to 23 months . twice: 2 doses with intervals between doses of at least 2 months;
- children aged 2 to 5 years once: 1 dose.
Contraindications:
- hypersensitivity due to previous vaccine administration;
- hypersensitivity to excipients and/or diphtheria toxoid;
- acute infectious and non-infectious diseases, exacerbation of chronic diseases (in these cases, vaccination is carried out after recovery or in remission).
Advantages:
- pneumococcal vaccine for newborns - helps protect the most vulnerable age group from the most common serotypes of pneumococcus, causing up to 80% of invasive pneumococcal infections;
- the presence of a population effect has been proven - a decrease in morbidity among unvaccinated children and adults due to the formation of stable collective immunity;
- One of the advantages of the drug is the binding of polysaccharides from the outer layer of the pathogenic bacterium to the “carrier” protein, which gives the vaccine permanent memory properties against many different types of pneumococci, including those that cause meningitis and pneumonia.
- Prevenar can be administered to children simultaneously (on the same day) with other vaccines included in the National Vaccination Schedule (with the exception of BCG), as well as with the vaccine against Hemophilus influenzae type b (Hib), according to the prescribed immunization schedule.
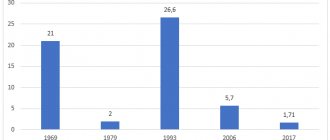
Application experience : since 2000, it has been registered and used in 90 countries of the world, 38 countries have included Prevenar in the National Childhood Vaccination Calendars, more than 260 million doses of the vaccine have already been administered in the world. Use of this vaccine in the United States has resulted in a significant reduction in the incidence of pneumococcal disease not only among immunized children but also among the nonimmunized population by reducing transmission. The effectiveness of the new vaccination is especially high in preventing infection in the family with inflammatory diseases of the pharynx and ears. The vaccine protects against 40% of all known types of pneumococci. The safety of the vaccine is high and side effects are rare. They are common - this is temperature, and local - this is swelling, temperature and pain at the injection site.
Registered in Russia since 2009.
How does the drug affect the immune system?
Vaccination is given to children starting from two months of age. They perform it strictly adhering to the prescribed scheme. This is necessary so that a constant flow of immune system reactions is created in the child’s body after each vaccine administered.
According to medical research, it has been shown that after the first vaccination in the child’s body, the level of antibodies increases. It is recommended to administer three doses of the pneumococcal drug Prevenar, observing the time period.
In children aged 2-5 years, after intramuscular administration of the drug, immunity to serotypes and strains of an infectious nature is observed. Their body response coincides with the group of children under two years of age.
During ongoing clinical trials, the Prevenar vaccine was found to be up to 97% effective against the pneumococcal group. The studies involved 18,000 children aged 2-15 months.
Features of the drug Prevenar
The main feature of this drug is its administration to children at an early age. According to the manufacturers' recommendations, the vaccine is indicated for children aged two months and older. This period of vaccination against pneumococcus has been approved and included in the National Vaccination Calendar.
Let's take a look at other differences between vaccines and other medications:
- The medicine is administered to children starting from two months and up to five years. Manufacturers believe that it is no longer advisable for adolescent children to receive such vaccinations;
- Adults, as well as adolescents, are not vaccinated with this drug;
- It is recommended to administer the medicine to children under two years of age only in the femoral part of the leg or in the gluteal muscle. After two years, the vaccine is injected into the shoulder surface - the deltoid muscle;
- The drug is administered three times, observing the required time interval. Revaccination is carried out for children aged one year and up to two years;
- If the child is not vaccinated according to schedule, then two doses of the vaccine must be administered at a time interval, and revaccination is carried out at two years. If the primary vaccination was given after two years, it is administered once.
Vaccination Prevenar after administration actively protects the child’s body from the effects of streptococcal infection, and also protects against complex consequences after suffering from the disease.
Is Prevenar vaccination mandatory or not?
Each state has its own list of mandatory vaccinations for children. In principle, it is not mandatory, but recently doctors have begun to require the Prevenar vaccine against pneumococcal infections to be administered before entering a primary care facility. The drug Prevenar 13 is given for a fee in private clinics, and in government institutions they give free vaccinations with an analogue of the drug Pneumo 23.
Actions before and after vaccination
To avoid side effects in a child after vaccination with Prevenar, you must adhere to the following simple recommendations:
- Before vaccination, you must visit your local pediatrician for an examination;
- If necessary, you need to get tested;
- After administering the medicine, there is no need to bathe the child or wet the injection site for 24 hours;
- If the child is breastfed, then the mother should not include exotic foods in her diet during this period.
Popular Why children need the Synflorix vaccine
If the weather conditions are good, then walking is not prohibited. But you need to be careful to have less contact with other children or sick adults for several days in order to avoid infection with any diseases.
"PREVENAR-13" - vaccination against pneumococcal infection
Pneumococcal polysaccharide vaccine, conjugated, adsorbed, thirteen-valent
Manufacturer: Wyatt Holdings Corporation, USA.
Protects against diseases: Pneumococcal infection.
Applicable: in children from 2 months onwards without age restrictions. Revaccination at 4, 6 and 12-15 months
- within the framework of the national calendar of preventive vaccinations;
- in individuals at increased risk of developing pneumococcal infection.
Included in the national vaccination calendar.
Advantages of the Prevenar 13 vaccine:
Prevenar 13 (Prevnar 13, PCV13, Pneumococcal, pneumococcal conjugate) is a high-quality vaccine produced in the USA by Wyeth Pharmaceuticals Inc. and registered in Russia, which helps protect a child from 13 strains of pneumococcal infection (Streptococcus pneumoniae strains 1, 3, 4 , 5, 6A, 6B, 7F, 9V, 14, 18C, 19A, 19F and 23F), causing diseases such as pneumonia, bronchitis, meningitis and sepsis.
- Invasive pneumococcal infection can cause brain damage or even death. Prevenar 13 can help protect children and is currently a safe and cost-effective method for preventing the development of infections. The vaccine is also approved for children 6 weeks to 5 years of age to prevent otitis media (an ear infection) caused by 7 of the 13 strains. The vaccine also stimulates the immunity of the mucous membranes, which leads to a decrease in nasopharyngeal bacterial carriage.
- Vaccination can be given to infants starting at 2 months of age (as directed by a doctor from 6 weeks) with booster vaccinations at 4, 6 and 12-15 months to create effective immune protection against the most common types of pneumococcal bacteria.
- Prevenar 13 is not 100% effective, but will only protect against the 13 strains included in the vaccine. It helps the body develop natural defenses, promoting the development of immunity to diseases. Like some other vaccines, it is given in 4 doses.
- Infants should receive 4 doses of the vaccine to help the baby's developing immune system develop effective, specific protection against pneumococci.
- Each subsequent vaccination is prescribed 4-8 weeks after the previous one, and the 4th dose cannot be prescribed earlier than 2 months after the 3rd vaccination. Children with weakened immune systems (eg, HIV infection, leukemia) may have a reduced immune response to vaccination.