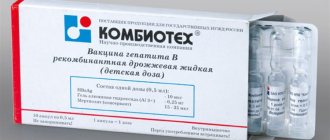
Hepatitis B vaccine - Vaccine against hepatitis B recombinant yeast liquid Registration number: P N000738/01 Trade name: against hepatitis B recombinant yeast liquid International nonproprietary or chemical name: against hepatitis B recombinant yeast liquid Dosage form: Suspensions for intramuscular administration Country of origin: Combiotech, Russia Preliminary examination: HbsAg, anti-HbsAg, if there is a history of hepatitis (if antibodies are present, vaccination is not necessary) Cost of the vaccine: 900 rubles (examination before vaccination is additionally paid) Applicable: For adults Dispensing conditions from pharmacies: only for medical and preventive and sanitary institutions Analog of this vaccine: “Hepatitis B recombinant yeast vaccine” (for children)
Leave a request for vaccination. Our manager will coordinate an appointment time convenient for you.
Sign up for vaccination
Sign up by phone
+7 (495) 788-000-2
DESCRIPTION OF THE MEDICINE
Hepatitis B recombinant yeast vaccine is a protein sorbed on aluminum hydroxide (HBsAg), synthesized by a recombinant yeast strain and containing antigenic determinants of the surface antigen of the hepatitis B virus.
Appearance: A homogeneous suspension of white, grayish tint, without visible foreign inclusions, which separates when standing into a colorless transparent liquid and a loose precipitate of white with a grayish tint, easily breaking when shaken.
Immunobiological properties
A course of vaccination leads to the formation of specific antibodies to the hepatitis B virus in a protective titer in more than 90% of vaccinated people.
Viral hepatitis B is an infectious liver disease caused by the virus of the same name, characterized by severe inflammatory damage to the liver. The disease has various forms - from viral carriage to acute liver failure, liver cirrhosis and liver cancer. Hepatitis B belongs to diseases transmitted in various ways. The most common are sexual contact and contact with blood infected with hepatitis B. You can become infected with hepatitis B when visiting a dentist, through a blood transfusion during preparation for surgery. By becoming infected yourself, a person puts his loved ones at significant risk.
Doctors who have frequent contact with blood - surgeons, dentists, laboratory workers - are also at great risk. In addition, the virus can be transmitted from a mother to her unborn child during pregnancy or directly during childbirth.
In Russia, over 50 thousand new cases of the disease are registered annually and there are over 5 million chronic carriers of the hepatitis B virus, who pose a danger as a potential source of infection. A large number of chronic carriers eventually develop a chronic form of the disease, progressing to cirrhosis and primary liver cancer. Hepatitis B virus is the main cause of one of the most common forms of cancer - primary liver cancer (hepatocellular carcinoma). The hepatitis B virus is reportedly 100 times more contagious than HIV.
The peculiarity of the hepatitis B virus is that it does not kill liver cells by itself. The disease, that is, hepatitis itself, is a consequence of the body's immune attack on its own liver cells. In principle, all routes of infection can be reduced to contact with biological fluids and human secretions. According to the degree of concentration of the virus in them, they can be arranged as follows: blood, semen, vaginal discharge, cervical discharge, tears, sweat, saliva, feces. The last four are less relevant for virus transmission. Infection does not occur through contact with saliva through kisses, cups, or musical instruments, but if there is an admixture of blood in the saliva, then infection is more than likely due to dental procedures or bites. The virus is also not transmitted by airborne droplets, through the bites of blood-sucking insects, to a child through mother's milk.
Based on this, we can list all possible ways of transmitting the virus: - transfusion of blood or its components:
- direct contact with contaminated blood - during blood transfusion,
- indirect contact with the patient’s blood (“syringe” route of infection, common among drug addicts, transmission through non-disposable medical instruments, combs, razors),
- sexual transmission (hetero- or homosexual contacts) - sexual transmission is dominant in countries with a good level of medical care. If during opposite-sex sexual contacts the virus is mainly transmitted with sperm and vaginal secretions, then during homosexual contacts - with sperm and blood from micro-tears and cracks. The risk of infection increases in proportion to the frequency of sexual contact with the source of the virus.
- transmission of the virus from mother to child - transfer of the virus from an infected mother to the fetus (so-called vertical transmission) occurs in utero (only 5% of cases) or directly during childbirth (95%), when the blood of the mother and child comes into contact directly, not through placental barrier. It is believed that if the virus was not transmitted to the child during childbirth, then infection, if the child is not vaccinated, will certainly occur over the next 5 years (so-called horizontal transmission). In the first year of a child's life, the virus can be transmitted from the mother through breast milk.
- household contacts with a sick or chronic carrier of the infection - The household route of transmission is especially relevant in families where there is a chronic carrier of the infection. It is believed that in this case, infection of all members of such a family necessarily occurs within 5-8 years.
The incubation period of hepatitis B lasts 45-180 days, with an average of 120 days. It is believed that from the moment of infection until the disease becomes inevitable, there is a period of 1-2 weeks, during which it is possible to prevent the disease through vaccination according to the 0-1-2-12 scheme ( emergency prophylaxis ) and the administration of immunoglobulin.
For children, hepatitis B poses a particular danger , since, if contracted at an early age, in 50-95% of cases it becomes chronic, which subsequently leads to liver cirrhosis and primary liver cancer. The younger the age at which infection occurs, the higher the likelihood of becoming a chronic carrier of the virus. In newborns, viral hepatitis is asymptomatic in 90-95% of cases, without classical jaundice and in 70-90% of cases leads to chronic carriage of the virus, and in 35-50% to chronic hepatitis.
In adults, these ratios are different - the frequency of diseases with jaundice is higher (30-40%), the risk of chronic infection is lower (6-10%). This dependence is associated with the degree of maturity of the immune system, because it has now been proven that the better the immune system works, the more pronounced the disease, with more symptoms, and the lower the risk of the infection becoming chronic.
Jaundice with hepatitis manifests itself in the form of yellowness of the whites of the eyes, yellowness of the skin, darkening of urine (to the color of relatively dark beer) and discoloration (to white) of feces. As a rule, jaundice is first detected by the patient's acquaintances by the change in the color of the whites of the eyes from white to lemon yellow. In some cases, jaundice begins with dark urine and discolored stool. In a third of cases, hepatitis B occurs as a flu-like illness , accompanied by loss of appetite and weakness, is not registered as hepatitis B and is detected after some time, when tested for the “Australian antigen”.
Jaundice is a positive signal indicating that immune system cells “see” and destroy infected liver cells (leading to bile entering the blood, etc.). The absence of jaundice indicates that the patient's immune system is not effective enough, and most likely the patient will become a chronic carrier. Children are more likely to become chronic carriers of the virus because their immune systems are less effective than those of adults.
Death rarely occurs after an acute illness and is observed only in rare, extremely severe fulminant forms of infection (the risk of which increases if the patient has chronic liver disease). Death is more likely to occur after 35-40 years, as a result of primary liver cancer, 60-70% of cases of which are a consequence of chronic hepatitis B.
Diagnosis of hepatitis B is based mainly on the identification of specific antigens of the virus. Based on which antigens are being detected at a given moment, one of the seven phases of the infection can be identified with a high degree of confidence.
Treatment for hepatitis B is expensive, often does not lead to a complete cure, and has numerous side effects.
The only reliable way not to get infected yourself and not to infect your loved ones is to get vaccinated.
All vaccines are intended for both children and adults, including risk groups, and are administered in age-specific doses intramuscularly. All vaccines are interchangeable. Since 2008, children of the 1st year have been vaccinated according to the 0-3-6 scheme, children in risk groups - according to the 0-1-2-12 scheme. Unvaccinated children, adolescents and adults are vaccinated according to the 0-1-6 schedule. After vaccination, antibodies can persist for up to 20 years or more; protection is also provided due to immunological memory. Therefore, WHO does not recommend revaccination, at least for 10-15 years; it is indicated only for health workers (every 7 years) and people at risk (hemodialysis, immunodeficiency).
INDICATIONS FOR USE
Prevention of hepatitis B in children within the framework of the national calendar of preventive vaccinations and persons from groups at increased risk of infection with the hepatitis B virus (children and adults in whose families there is a carrier of HBsAg or a patient with chronic hepatitis B; children of orphanages, orphanages and boarding schools; children and adults regularly receiving blood and its preparations, as well as those on hemodialysis and oncohematological patients; persons who have had contact with material infected with the hepatitis B virus; medical workers who have contact with blood; persons involved in the production of immunobiological preparations from donor and placental blood ; students of medical institutes and students of secondary medical educational institutions (primarily graduates); persons who use injecting drugs). In addition to the above categories, all other groups of the population should be vaccinated.
Vaccination schedules
For adults
Adults who have not previously been vaccinated against hepatitis B are vaccinated as follows:
| Vaccine introduction | Deadlines |
| First time | when applying to a medical institution for registration |
| Second time | 1 month after the first vaccination |
| Third time | 6 months after the second vaccination |
If there are patients with hepatitis B in the family or the patient has close contact with other infected people, then the following scheme is used:
| Vaccine introduction | Deadlines |
| First time | when applying to a medical institution for registration |
| Second time | 1 month after the first vaccination |
| Third time | 1 month after the second vaccination |
| Fourth time | 10 months after the third vaccination |
This scheme is also relevant for children who often come into contact with people infected with hepatitis B.
For children
Children whose mother is infected with hepatitis B or has not been vaccinated at one time are vaccinated according to the following scheme:
| Vaccine introduction | Deadlines |
| First time | within 24 hours after birth |
| Second time | when the baby turns one month old |
| Third time | at the age of two months |
For other children, the following scheme is used:
| Vaccine introduction | Deadlines |
| First time | within 24 hours after birth |
| Second time | at one month of age |
| Third time | at the age of 6 months (usually given simultaneously with the whooping cough-diphtheria-poliomyelitis-tetanus vaccine) |
The same scheme is used for children who were not vaccinated in the maternity hospital. That is, the first vaccination is carried out when adults approach the child, the second is carried out a month later, the third after 6 months.
If after the first vaccination negative symptoms were observed, a rise in temperature and the person did not tolerate it well, then subsequent vaccinations should be done only after consultation with a specialist.
METHOD OF APPLICATION AND DOSAGE
The vaccine is administered intramuscularly: to adults, adolescents and older children in the deltoid muscle, to newborns and young children in the anterolateral part of the thigh. Administration to another site is undesirable due to a decrease in the effectiveness of vaccination. Do not administer intravenously. Children of the first year of life, as well as those with a history of various somatic diseases, are recommended to be vaccinated using a vaccine that does not contain a preservative (merthiolate).
A single dose is:
- For persons over 19 years of age - 1 ml (20 mcg HBsAg);
- For children and adolescents up to 19 years of age inclusive - 0.5 ml (10 mcg HBsAg);
- For patients in the hemodialysis department 2 ml (40 μg HBsAg).
The vaccine in ampoules containing 1 ml of the drug (adult dose) can be used to vaccinate 2 children (0.5 ml each) provided they are vaccinated simultaneously.
Application note:
- Shake the vaccine before use;
- For injection, use only a disposable syringe;
- The opening of ampoules and the vaccination procedure are carried out in strict compliance with the rules of asepsis and antiseptics;
- The injection site is treated with 70% alcohol before and after the injection;
- The drug cannot be stored in an opened ampoule.
Vaccination of children as part of the national calendar of preventive vaccinations
Vaccination against viral hepatitis B is carried out for all newborns in the first 24 hours of the child’s life. Newborns from risk groups are vaccinated according to the 0-1-2-12 schedule (the first dose - in the first 24 hours of the child’s life, the second at the age of 1 month, the third dose - at the age of 2 months, the fourth dose - at the age of 12 months). Simultaneously with the first vaccination, it is recommended to inject human immunoglobulin against hepatitis B intramuscularly into the other thigh at a dose of 100 IU. The same scheme is used to vaccinate children at risk who were not vaccinated in the maternity hospital for medical reasons after the latter have been removed.
Newborns and all children of the first year of life who are not at risk are vaccinated according to the 0-3-6 schedule (1 dose at the start of vaccination, 2 dose - 3 months after 1 vaccination, 3 dose - 6 months after the start of immunization) .
Children who have not received vaccinations under the age of 1 year and who do not belong to risk groups, as well as adolescents and adults who have not been vaccinated previously, are vaccinated according to the 0-1-6 schedule (1 dose at the start of vaccination, 2 dose - 1 month after 1 vaccination, 3 dose – 6 months from the start of immunization).
Note for use in children within the national immunization schedule:
- If the interval between the first and second vaccinations is extended to 5 months or more, the third vaccination is carried out 1 month after the second;
- Vaccinations of persons who have had contact with material infected with the hepatitis B virus are carried out according to the 0-1-2 month schedule. Simultaneously with the first vaccination, human immunoglobulin against hepatitis B should be administered intramuscularly (to another place) at a dose of 100 IU (children under 10 years old) or 6-8 IU/kg (other ages). In these individuals who have previously received a full course of vaccination against hepatitis B, the content of antibodies to HbsAg is determined before the second vaccination. If antibody titers are at least 100 IU/l, the second and third vaccinations are not carried out;
- Persons at risk, due to their professional characteristics, who have constant contact with blood, should be tested annually for antibodies to HbsAg. If the antibody titer decreases below 100 IU/l, revaccination with one dose of the vaccine is recommended;
- For unvaccinated patients scheduled for surgery, an emergency vaccination schedule of 0-7-21 days is recommended;
- For patients in the hemodialysis department, the vaccine is administered four times according to the scheme 0-1-2-6 months.
Vaccine hepatitis B recombinant (rDNA)
Before use, the vial (ampoule) with the vaccine must be shaken well several times until a homogeneous suspension is obtained.
The vaccine is administered intramuscularly:
- for young children (1-2 years) - in the upper outer surface of the middle part of the thigh;
- adults, teenagers and older children (over 2 years old) - into the deltoid muscle.
For patients with bleeding disorders, the vaccine should be administered subcutaneously.
It is prohibited to administer the vaccine intravenously!
When administering the vaccine, you should make sure that the needle does not enter the vascular bed. The drug from an opened bottle with 10 doses of the vaccine must be stored at a temperature of 2-8 0 C and used within one day.
A single dose of the vaccine is:
— for children from 1 year old, teenagers and persons under 19 years old
- 0.5 ml (10 µg HBsAg);
— for persons over 19 years old
- 1 ml (20 µg HBsAg).
Vaccination against viral hepatitis B for persons not previously vaccinated and not at risk
, is carried out in accordance with the National calendar of preventive vaccinations of the Russian Federation and the calendar of preventive vaccinations for epidemic indications (Order of the Ministry of Health of Russia dated March 21, 2014 No. 125n) according to the 0-1-6 scheme (1st dose at the start of vaccination, 2nd dose - 1 month after the 1st dose, 3rd dose - 6 months after the 1st dose).
Children at risk
(born from mothers who are carriers of HBsAg, patients with viral hepatitis B or who have had viral hepatitis B in the third semester of pregnancy, who do not have test results for markers of hepatitis B, who consume narcotic drugs or psychotropic substances, from families in which there is a carrier of HBsAg or a patient with acute viral hepatitis B and chronic viral hepatitis) vaccination is carried out according to the 0-1-2-12 scheme (1st dose at the start of vaccination, 2nd dose 1 month after the 1st dose, 3rd dose after 2 months after the 1st dose, 4th dose - 12 months after the 1st dose).
Contact persons from the outbreaks of the disease who have not been sick, have not been vaccinated and do not have information about preventive vaccinations against viral hepatitis B are subject to vaccination according to the 0-1-6 scheme.
Vaccinations against hepatitis B according to the 0-1-6 scheme are also subject to:
- children and adults who regularly receive blood and its preparations;
— oncohematological patients;
- medical workers who have contact with the blood of patients;
— persons involved in the production of immunological preparations from donor and placental blood;
— students of medical institutes and students of secondary medical educational institutions (primarily graduates);
— persons who inject drugs.
Patients receiving hemodialysis treatment
, the vaccine is administered four times according to the scheme: 0-1-2-6 or 0-1-2-3 at a double age dose
Unvaccinated persons who have been exposed to hepatitis B virus-infected material should
vaccination is carried out according to the 0-1-2 scheme. Simultaneously with the first vaccination, it is recommended to administer intramuscularly (to another place) human immunoglobulin against hepatitis B at a dose of 100 IU (children under 10 years old) or 6-8 IU/kg (other ages).
Unvaccinated patients scheduled for surgery
, it is recommended to vaccinate according to the 0-7-21 day schedule one month before surgery.
SIDE EFFECTS
Side effects with the vaccine are rare. In 1.8-3.0% of cases, slight transient pain, erythema and induration at the injection site are possible, as well as a slight increase in temperature, complaints of malaise, fatigue, joint pain, muscle pain, headache, dizziness, nausea.
These reactions develop mainly after the first two injections and disappear after 2-3 days.
Considering the possibility of the extremely rare development of immediate allergic reactions in particularly sensitive individuals, vaccinated persons must be provided with medical supervision for 30 minutes.
Vaccination sites must be provided with anti-shock therapy.
Vaccine prevention of hepatitis B
Hepatitis B is one of the ubiquitous infectious diseases and is no less a problem of the 20th century. than AIDS. In terms of prevalence, hepatitis exceeds the “plague of the century” by thousands of times. Hepatitis B virus (HBV) is an acute or chronic liver disease caused by a DNA (deoxyribonucleic acid)-containing virus transmitted parenterally. It is characterized by a slow development of the disease, a long course, significant spread, severe and in some cases fatal outcomes, a high level of transition to a chronic form with further outcome in cirrhosis and/or primary liver cancer. HBV remains an important cause of morbidity and mortality worldwide (about 50 million patients with acute HBV alone are reported annually). Of these, up to 600 thousand patients with HBV die. In the United States alone, there are approximately 1–1.5 million infected people, the annual increase in infected people is from 10 to 15 thousand people, and about 5.5 thousand patients die annually from the consequences of HBV. In Russia, the epidemic situation regarding HBV also remains very serious - over 50 thousand new cases of the disease are recorded annually. The prevalence of HBV varies significantly by region. For example, in the Novosibirsk region, the incidence rate among people aged 20–29 years is 40.0 per 100 thousand people and tends to decrease after 30 years (the incidence rate among people 30–39 years old is 25.1 per 100 thousand people). ). The spread of injecting drugs among young people (40–60% of all cases of parenteral viral hepatitis are drug addicts infected through intravenous drug use), a significant increase in the number of promiscuous sexual contacts explain the involvement of young people of working age in the epidemiological process. Young people under 30 years of age who used drugs account for 80% of those who die from HBV. More than 1/3 of deaths are caused by simultaneous infection with hepatitis B, C and D viruses. Another feature of HBV is the widespread distribution of virus carriage. According to WHO estimates, there are over 350 million such people around the globe. Young people are at greater risk of developing chronic HBV carriage and associated pathology than older people. Among “healthy” carriers, there is a significant percentage of undetected asymptomatic forms of infection.
The only source of HBV infection is a person with an acute or chronic form of the disease, as well as, to a greater extent, a “healthy” virus carrier. HBV spreads through evolutionarily formed natural and artificial transmission routes. Artificial transmission routes have been studied quite widely. Of great importance, along with blood transfusions, are the increasing volumes and types of parenteral medical and non-medical manipulations. The risk group for HBV infection includes medical workers who, in the course of their professional activities, come into contact with blood and its preparations (surgeons, dentists, hemodialysis workers, laboratories, etc.). At the same time, the share of nosocomial morbidity in the structure of the general morbidity of the population decreases annually.
Artificial routes of transmission of the virus include non-medical manipulations such as parenteral drug administration, earlobe piercing, tattooing, manicure, shaving, and cosmetic procedures if they are performed with instruments that have not been reliably processed.
Natural routes of transmission of the virus include transmission of the virus through sexual contact, vertical transmission from mother to child in the second half of pregnancy when the placental barrier is broken (a prerequisite for such transmission of the virus is maternal illness), various types of direct and indirect contact in everyday life.
For example, data on the routes of transmission of HBV in patients with an acute form of infection in Moscow in 1999 are as follows:
- sexual - 51.5%;
- parenteral drug administration - 30.1%;
- unidentified - 10.1%;
- contact and household - 4.9%;
- infection in medical institutions in Moscow - 3.3%;
- vertical - 0.1%.
Consequently, in modern conditions, HBV infection mainly occurs through the blood. More often this happens when using shared syringes for intravenous drug administration, as well as toothbrushes and razors, when performing tattoos, piercings, manicures, and less often when removing teeth or conducting examinations that violate the integrity of the skin or mucous membranes. Infection during operations is also possible. In other words, the virus can enter the body through piercing objects and medical instruments, which, if not properly treated, retain particles of infected blood. Transfusion of blood containing the virus can also cause infection. If a sexual partner has HBV, then the probability of transmission of infection through semen is 30%; the virus penetrates even through intact mucous membranes. It is also found in saliva, tears, urine and even feces; if they come into contact with damaged skin and mucous membranes, there is a risk of infection, but it is very small. Such transmission of the virus is possible in everyday life, most often among children. Cases of hepatitis infection have been identified when using a single tube for inhaling cocaine. If a mother has HBV, she may give birth to an infected child. The virus is found in mother's milk, and there is no risk of infection for the baby; breastfeeding is allowed. However, in newborns, HBV usually becomes chronic (90%), when infected in the first year of life - in 50% of cases, in adults - in 5-10%.
After infection, the incubation period lasts from 42 to 180 days, with an average of 60–120 days. In half of the cases, the disease begins with signs of a mixed version of the pre-icteric period, occurring with normal or subfebrile body temperature. Symptoms of intoxication and dyspeptic manifestations are moderate. In 30–35% of patients in the initial period, an arthralgic variant of the disease is observed, a feature of which is increased pain in large joints at night and in the morning. Another 10–12% of patients may develop urticarial skin rashes that persist for 1–2 days and are accompanied by eosinophilia in the peripheral blood. In 5–7% of cases, signs of intoxication are completely absent, and icterus of the sclera and skin, darkening of the color of urine may be the first clinical manifestations of the disease. The initial period of illness lasts 7–14 days or more, but for infections associated with blood transfusion, it may be shorter.
During the icteric period, which lasts 3–4 weeks, persistent clinical symptoms appear. As a rule, against the background of weakness and loss of appetite, pain and sometimes quite sharp pain occur in the right hypochondrium. Nausea and even vomiting are common. Often (in almost 20% of cases) itching of the skin is noted. The liver is always enlarged, smooth on palpation, with a somewhat compacted consistency. Possible enlargement of the spleen. Gallbladder symptoms may be positive. The hemogram most often reveals leukopenia with lymphocytosis and monocytosis, sometimes with a plasmatic reaction. The erythrocyte sedimentation rate is reduced to 2–4 mm/h; during the period of convalescence it can accelerate to 18–24 mm/h with subsequent normalization in the absence of complications. Hyperbilirubinemia is usually severe and persistent, especially in the second half of the icteric period. As a rule, there is a manifest increase in the activity of aminotransferases in the blood serum with a decrease in the sublimate test and prothrombin index. A serological blood test reveals HbsAg (surface antigen), anti-HBc immunoglobulin (Ig)M (antibodies to the core antigen).
In protracted forms of HBV, clinical and biochemical manifestations at the height of the disease and especially during its reverse development are long - from 3 to 6 months. These forms may be a prestage of chronic hepatitis. It has been established that hematogenous circulation in a consistently high concentration of HBV DNA for more than 3 weeks, HbeAg (nuclear antigen polypeptide) for more than 1 month, HBsAg for more than 3 months, anti-HBc IgM characterizes the progradient course of HBV, and, accordingly, more than 5 weeks, 2 and 6 months predicts likelihood of chronicity. Chronic hepatitis occurs in 5–10% of adults.
A preliminary diagnosis of acute HBV is made if the patient, 1.5–6 months before the onset of the disease, received blood, plasma, erythrocyte, leukocyte, or platelet transfusions, surgical interventions, endoscopic examinations, numerous injections (including drugs), or what happens much less frequently if the patient has had sexual or close contact with a patient with HBV. Clinically manifest acute HBV is characterized by a gradual onset of the disease, a long pre-icteric period with polyarthralgia and possible allergic skin rashes, no improvement in health or its deterioration with the appearance of jaundice, a long icteric period with a slow disappearance of symptoms of the disease during the period of convalescence. To confirm the diagnosis, detection of HBsAg, HBeAg, anti-HBc IgM, as well as HBV DNA in the blood is necessary. The favorable cyclical course of hepatitis corresponds to the rapid disappearance of first HBeAg with the appearance of anti-Hbe, HBV DNA, then HBsAg with the appearance of anti-HBs. Early anti-HBc IgM is replaced by late anti-HBc IgG. One should also think about the possible development of chronic hepatitis when HBsAg is detected in a stable titer for 6 months or more from the onset of the disease.
In the current conditions of high prevalence of HBV, in order to reduce the level of morbidity and virus carriage, it is necessary to introduce modern preventive technologies. The introduction of the principles of a healthy lifestyle can be considered as the most optimal and adequate primary prevention measure aimed at the entire population.
In the preventive complex, a special place is given to measures aimed at preventing HBV infections during blood transfusions and carrying out therapeutic and diagnostic parenteral procedures. The introduction of autohemotransfusions, when a patient is transfused with his own blood, collected before a planned operation, is a promising direction.
Nonspecific prevention of HBV involves the use of disposable instruments for manipulations associated with damage to the skin and mucous membranes, thorough sterilization of medical instruments, as well as strict control to detect viral antigens in donors. It should be borne in mind that the hepatitis B virus is extremely stable in the external environment. In whole blood and its components it persists for years. The antigen is detected on bedding, medical and dental instruments, and needles contaminated with blood serum for several months at room temperature. Transmission of the virus can be avoided by observing the following sanitary and hygienic rules: individualization of all personal hygiene items and their separate storage (razors, toothbrushes, washcloths, combs, etc.), compliance with personal hygiene rules, prevention of microtraumas in everyday life and at work.
To prevent sexual transmission of infection, casual sexual intercourse should be avoided; It is also recommended to use mechanical contraceptives.
Prevention of occupational infections in medical institutions is achieved by strict adherence to the rules of the anti-epidemic regime, especially in hemodialysis, surgical, laboratory and other departments where personnel often have contact with blood. When performing any parenteral interventions and procedures, the use of rubber gloves is mandatory.
From the standpoint of evidence-based medicine, given the variety of routes of transmission of HBV and the large number of sources of infection, the most promising means of prevention is vaccination. This is the only way to prevent HBV in newborns. Vaccination prevents the development of liver cancer and reduces the level of carriage of the virus in the population. Today there is every reason to classify HBV as a vaccine-preventable infection, the spread of which can be controlled using specific preventive measures [7, 16].
There is now sufficient evidence to suggest that eradication of HBV is possible this century through extensive vaccine prevention programs.
Over the past decade, more than 150 million people have been vaccinated in the world, which has not only made it possible to sharply reduce the incidence of HBV and significantly influence the level of carriage in the population of a number of countries, but also to make it legitimate to raise the issue of complete eradication of HBV in these countries. More than 75 countries have included routine HBV immunization of newborns and/or adolescents in their vaccination programs. Russia is also included in these countries—vaccination against HBV is now included in the national vaccination calendar.
Currently, genetically engineered and plasma vaccines are used (the latter are not licensed in the Russian Federation). Genetically engineered vaccines are produced using recombinant technology by incorporating the gene for the S subunit of the hepatitis B virus, encoding HBsAg, into the genome of yeast cells. HBsAg synthesized by yeast is highly purified from yeast proteins. Below are the most commonly used yeast recombinant vaccines in Russia (Table 1).
These vaccines induce the formation of specific antibodies to HBsAg (anti-HBs antibodies). An anti-HBs antibody titer of 10 IU/L is sufficient to create immunity against HBV. Available evidence suggests that achieving these antibody levels after primary immunization results in the formation of long-term immunological memory and provides continued protection against HBV even if antibody levels subsequently decline.
Since the beginning of the 90s of the last century, a sufficient amount of data has been accumulated in world medical practice indicating the high immunogenic and preventive effectiveness of three-time immunization of healthy children with the Euvax B vaccine. Administration of a dose equal to 10 mcg to healthy newborns, as well as preschool and school-age children according to The standard regimen provides a 94–100% seroconversion rate with an average antibody blood concentration of 874.4 mIU/ml [11].
According to Russian researchers [11], the low reactogenicity of the vaccine was revealed during vaccination among schoolchildren in Podolsk and Moscow. Among the noted post-vaccination reactions, mild local reactions were observed in the form of redness at the injection site, and general reactions included a rise in body temperature to 37.5 °C. A study of the intensity of the immune response showed the presence of anti-HBs in 99% of vaccinated people. At the same time, 80.3% of children had a high concentration of specific antibodies in the blood (above 1000 IU/l).
The vaccination course is carried out according to two main schemes: the first scheme, which provides vaccination at monthly intervals (0-1-2 months), ensures a more rapid increase in antibodies and is recommended for emergency prophylaxis (newborns from mothers who are carriers of HBsAg) and in cases of possible HBV infection with carrying out surgical intervention and other parenteral manipulations, as well as laboratory tests. With this regimen, revaccination is carried out after 12–14 months. When vaccinated according to the second scheme (0-1-6 months, i.e., the first two injections are given at an interval of 1 month and the third - 6 months after the start of vaccinations), the immune response is developed more slowly, but a higher antibody titer is achieved.
All vaccines are intended for immunoprophylaxis of both children and adults, including risk groups.
According to Order of the Ministry of Health and Social Development of the Russian Federation No. 229 dated June 27, 2001 “On the national calendar of preventive vaccinations and the vaccination calendar for epidemic indications,” persons in risk groups are subject to vaccination regardless of age:
- children and adults, previously unvaccinated, whose families have a carrier of HbsAg or a patient with chronic HBV;
- children from children's homes, orphanages and boarding schools;
- children and adults who regularly receive blood and its preparations, as well as those on hemodialysis and oncology-hematological patients;
- persons who have had contact with HBV-infected material;
- medical workers who come into contact with the blood of patients;
- persons involved in the production of immunobiological preparations from donor and placental blood;
- students of medical institutes and students of secondary medical institutions (primarily graduates);
- persons who use drugs and inject drugs.
In the current epidemiological situation, it is advisable to vaccinate homosexual men, drug addicts and prisoners, whose risk of infection is especially high.
Vaccines against HBV can prevent infection early after infection. An example is the vaccination of newborns from carrier mothers. In a number of cases (emergency prophylaxis for unvaccinated people, passive immunization of immunocompromised patients, etc.), specific immunoglobulins are used (Table 2).
It is well known that the tactics of selective vaccination of only risk groups, previously carried out both in our country and abroad, are ineffective and do not provide a significant level of morbidity reduction. To achieve a significant reduction in the incidence of HBV, it is necessary to move to mass vaccination of the entire population.
In conclusion, it should be noted once again that in Russia the HBV problem is aggravated by the rapid increase in the incidence of HBV and mixed infections of viral hepatitis B, C and D among young people due to the increase in intravenous drug use. Among patients, there is a tendency to increase the frequency of chronic viral hepatitis. Until now, the efforts made to solve this problem by medical organizations and the financial contribution of society remain inadequate, taking into account the real threat to public health from the spread of viral hepatitis and the onset of their dangerous consequences.
According to experts, in our country the first and most important task is mass vaccination against HBV of newborns and adolescents not yet infected with HBV. In clinical practice, the most pressing tasks for infectious disease specialists remain the development and provision of technology for early prediction of the development and chronicity of severe HBV, the creation of highly effective means and procedures to prevent complications of infection and intensive treatment of severe patients.
For all specialists involved in providing medical care to hepatitis patients, daily practical mastery of new incoming information about viral hepatitis, familiarization with new methods and means of diagnosing and treating HBV and other viral hepatitis are important and relevant.
Literature
- Balayan M. S. Hepatitis A: yesterday, today, tomorrow // Medicine for everyone. 1999. No. 2. P. 22-25.
- Blyuger A.F., Novitsky I.N. Viral hepatitis. Riga: Zvaigzne, 1988. 414 p.
- Vinogradova E. N., Yakovlev A. A., Demidenko T. P. About some causes of protracted forms of viral hepatitis A // Clinical medicine. 1996. No. 9. pp. 29–31.
- Gubergrits N. B. Chronic hepatitis and cirrhosis of the liver. Modern classification, diagnosis and treatment. Donetsk: LLC Lebed, 2002. 156 p.
- Diagnosis and treatment of chronic viral hepatitis B, C and D in children // Scientific and practical program for doctors. M., 2002. 60 p.
- Zhdanov K.V. Latent forms of viral hepatitis B and C in young people: abstract. dis. ...Dr. med. Sci. St. Petersburg, 2000. 44 p.
- Kaganov B. S. Viral hepatitis B: achievements and problems // Russian Pediatric Journal. 1998. No. 1. P. 50–60.
- Kozhemyakin L. A., Ketlinskaya O. S., Romanova S. Yu. New opportunities in the treatment of viral hepatitis // Attending physician. 2001. No. 1. P. 34–35.
- Kramarev S. O. Infections and diseases in children (clinical lectures). K.: Morion, 2003. 479 p.
- Kuzin S.N. // Vaccination. 2001. No. 3 (15). pp. 8–9.
- Lobzin Yu. V., Zhdanov K. V., Volzhanin V. M. Viral hepatitis. St. Petersburg: Foliant, 1999. 104 p.
- Mayer K. Hepatitis and consequences of hepatitis. M.: Geotar Medicine, 1999. 423 p.
- Guide to infectious diseases / ed. prof. Yu. V. Lobzina. St. Petersburg: Foliot, 2000. 936 p.
- Tatochenko V.K., Ozeretskovsky N.A., Immunoprophylaxis: (reference book - 6th ed., add.). M., 2003. P. 174.
- Shakhgildyan I.V., Khukhlovich P.A., Mikhailov M.I. et al. Results of 10 years of use of the Engerix B vaccine for the prevention of HB virus infection in Russia. Research Institute of Virology named after. D. I. Ivanovsky RAMS, Research Institute of Epidemiology and Microbiology named after. N. F. Gamaleyi RAMS, Federal, Moscow City and Sverdlovsk Regional Centers of State Sanitary and Epidemiological Supervision, 2001.
- Sherlock S., Dooley J. Diseases of the liver and biliary tract: practical work. manual / translation from English M.: Geotar Medicine, 1999. 864 p.
- Cherednichenko T.V., Moskovskaya I.A. Viral hepatitis in children of the first year of life // Children's infections. 2003. No. 3. P. 11-13.
- Alter MJ Epidemiology of hepatitis C// Hepathology. 1997; 26:62–65.
- Andre F., Coates T., Wilson R., Patrick G., Watson V. Meta-analysis of published clinical studies on two yeast-derived hepatitis B vaccines to determine attainable seroprotection rates // Antiviral therapy. 2000; 5 (Suppl. 1): B45, abstract B082.
- Assad S., Francis A. Over a decade of experience with a yeast recombinant hepatitis B vaccine // Vaccine. 2000; 18: 57-67.
- Banatvala J., Van Damme P., Oehen S. Lifelong protection against hepatitis B: the role of vaccine immunogenicity in immune memory// Vacine. 2001; 19:877–885.
- Coates T., Wilson R., Patrick G., Andre F., Biol MI, Watson V. Hepatitis B vaccines: assessment of the seroprotective efficacy of two recombinant DNA vaccines // Clin Ther. 2001; 23(3): 392-03.
- Demir H., Saltic-Temisel N., Usta Y. et al. Long-term results of interferon (treatment in children with chronic hepatitis B// J. Hepatology. 2005; 42 (Suppl. 2.): 174.
- Dienstag JL, Schiff ER, Wright TL et al. Lamivudine as initial treatment for chronic hepatitis B in the United States // N. Engl. J. Med. 1999; Vol. 341, No. 17: 1256–1263.
- Leroux-Roels G., Abraham B., Fourneau M., De Clercq N., Safari A. A comparison of two commercial recombinant vaccines for hepatitis B in adolescents Vaccine 2001 (19): p. 937–939.
- Minola E. Prati D Age at infection affects tie long-term outcome of transfusion-associated chronic hepatitis C// Blood. 2002; 99: 4588-4591.
- Naoumov N.V., Schneider R., Grotzinger T. et al. Precore mutant hepatitis B virus infection and liver disease // Gastroenterology.1992; Vol.102, N2.: 538–543.
- Vryheid RE, Kane MA, Muller N., Schatz GC, Bezabeh S. Infant and adolescent hepatitis B immunization up to 1999: a global overview // Vaccine. 2001; 19: 1026–1037.
A. B. Malakhov , Doctor of Medical Sciences, Professor MMMA named after. I. M. Sechenova, Moscow
CONTRAINDICATIONS
- Hypersensitivity to yeast and other components of the vaccine;
- Severe reaction (temperature above 40 C, swelling, hyperemia >8 cm in diameter at the injection site) or complication (exacerbation of chronic diseases) to the previous administration of the drug. Routine vaccination is postponed until the end of acute manifestations of the disease or exacerbation of chronic diseases. For mild ARVI, acute intestinal and other diseases, vaccinations can be carried out after the temperature has normalized.
CLAIMS
All cases of increased reactogenicity or development of post-vaccination complications should be reported to:
- Federal Service for Supervision of Consumer Rights Protection and Human Welfare;
- The Ministry of Health and Social Development of Russia and the Federal State Institution GISC named after. L.A. Tarasevich Rospotrebnadzor with subsequent sending of medical documentation: 121002, Moscow, lane. Sivtsev - Vrazhek, 41, tel., fax +7;
- Manufacturer's address: JSC NPK "Combiotech", 117997, Moscow, Miklukho-Maklaya str., 16/10, building 71, tel./fax +7 (495) 330-74-29.